Metformin and LFS
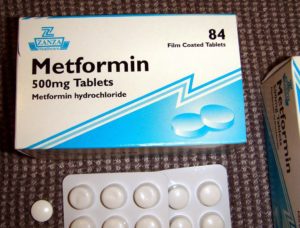
What is Metformin? Metformin is an oral medication that decreases the amount of sugar made in the liver, helps the body use sugar and recognize

What is Metformin? Metformin is an oral medication that decreases the amount of sugar made in the liver, helps the body use sugar and recognize
One of the most exciting presentations at the REACH 16 International LFS Conference was information presented on COTI-2, a novel small molecule that helps repair
One of the big complaints with rare syndromes is that there aren’t enough clinical studies to address our needs. PROMPT is a study that combines
The day was warm, which was quite surprising because Bethesda had just gotten 7 inches of snow the day before. My flight out of Greensboro
The NIH recently released information regarding a new Clinical Trial for people with LFS. The Trial(#NCT01981525) uses Metformin, a drug traditionally used to treat type
Magnetic Resonance Imaging. MRI. MRIs use powerful magnets and radio waves to create images of the body. The benefit to people with LFS is that